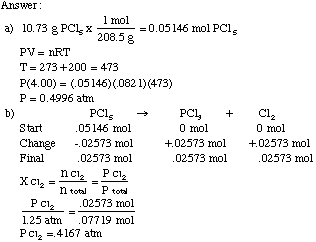
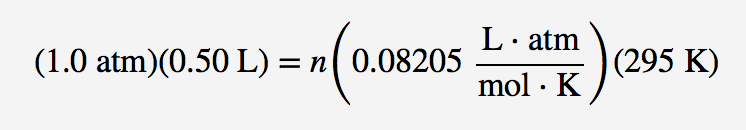Solved] Trial Volume Pressure Temperature Moles Concentration Moles Density R Kb (mL) (atm) (K) of Gas of Gas of gas of Gas (atm (atm (moles/L) (gra... | Course Hero

A 16.0 L gas cylinder is filled with 7.60 moles of gas. The tank is stored at 27^oC. What is the pressure in the tank? | Socratic