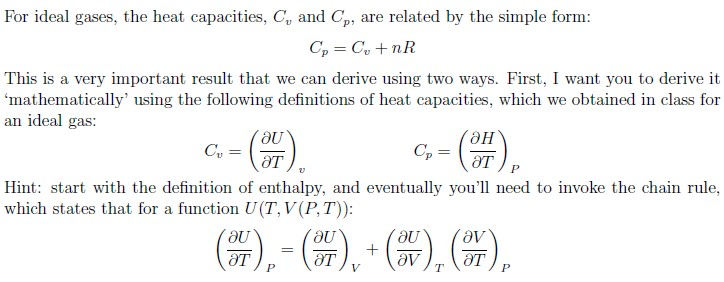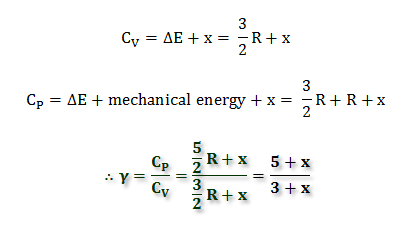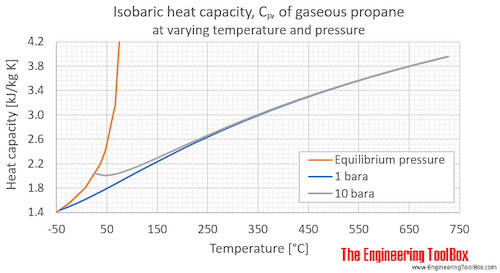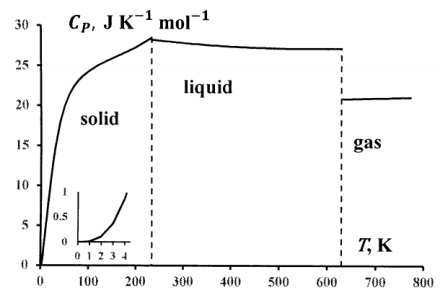
Variation of Natural Gas Heat Capacity with Temperature, Pressure, and Relative Density | Campbell Tip of the Month
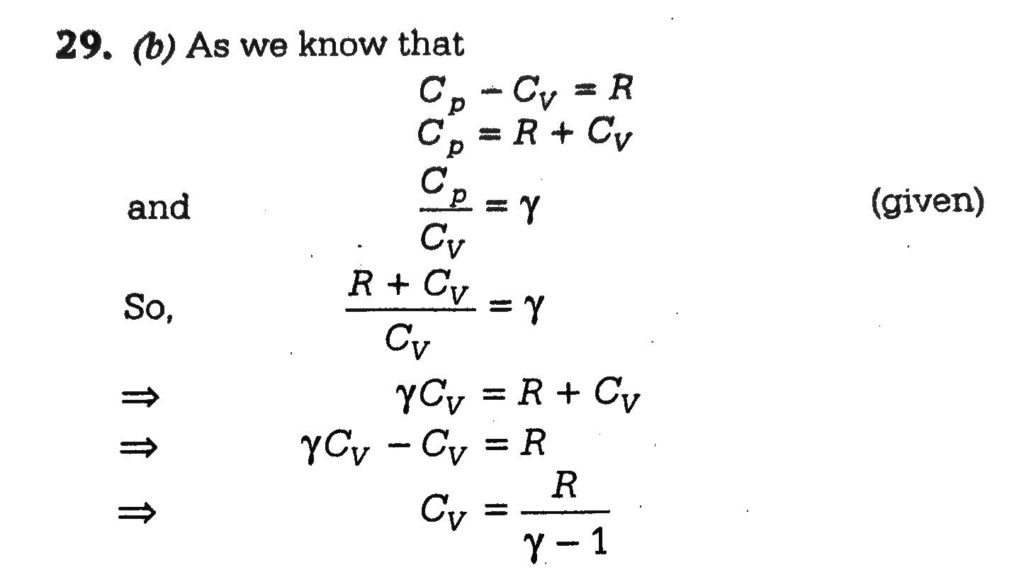
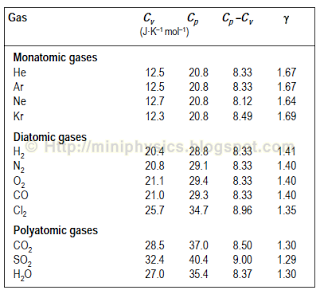
The molar specific heats of an ideal gas at constant pressure and volume are denoted by Cp and Cv respectively. If γ=Cv/Cp and R is the universal gas constant, then Cv is
Variation of Natural Gas Heat Capacity with Temperature, Pressure, and Relative Density | Campbell Tip of the Month

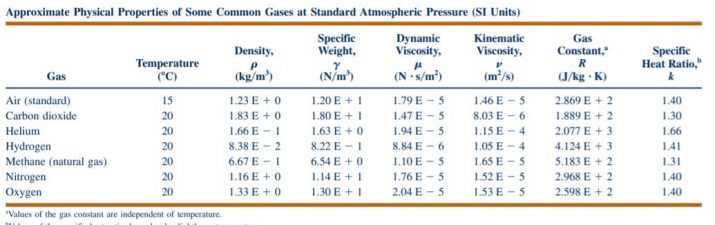
A new approach for simplifying the calculation of flue gas specific heat and specific exergy value depending on fuel composition - ScienceDirect

An ideal gas (Cp/Cv = y) is taken through a process in which the pressure and the volume vary as P = aV^b. Find the value of b for which the specific

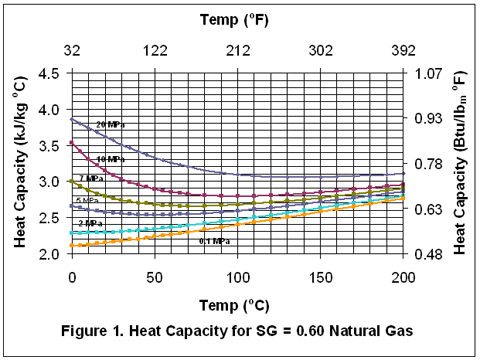
Variation of Ideal Gas Heat Capacity Ratio with Temperature and Relative Density | Campbell Tip of the Month

A new approach for simplifying the calculation of flue gas specific heat and specific exergy value depending on fuel composition - ScienceDirect

Calculate the specific heat of a gas at constant volume from the following data. Desity of the gas - YouTube