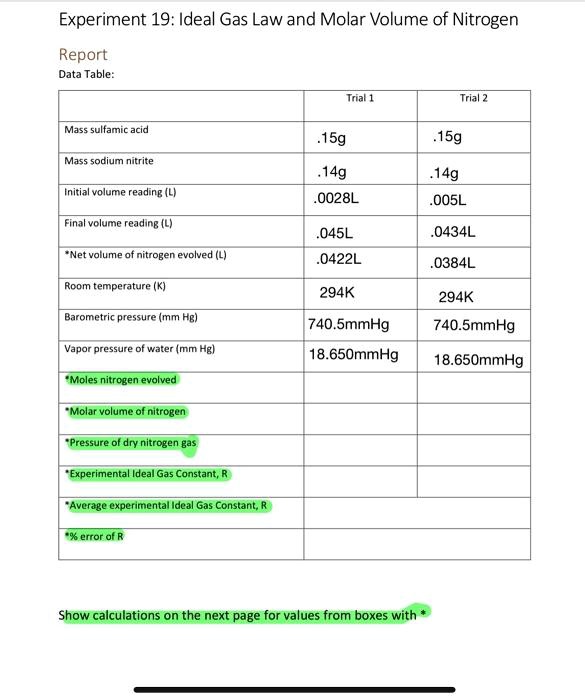
SOLVED:Experiment 19: Ideal Gas Law and Molar Volume of Nitrogen Report Data Table: Inal 1 Trial 2 Mass sulfamic acid 15g .15g Mass sodium nitrite 14g 0028L 14g OO5L Inmtalvolume reading (L

A dry gas occupies 224 cm3 at a normal pressure. If its volume increases by 25%, find the new - YouTube

thermodynamics - why sometimes we can find p=ρgRT instead of p=ρRT for ideal gas? - Physics Stack Exchange
For air, the characteristic gas constant R = 0.287 kJ/ kg.K [17] Question 4 Ammonia gas enter the reactor of a nitric acid plant with 30 % more dry... | Course Hero

The Ideal Gas Law. Gas Constant. Applications of the Gas law. P = ρ R T. Lecture 2: Atmospheric Thermodynamics - PDF Free Download