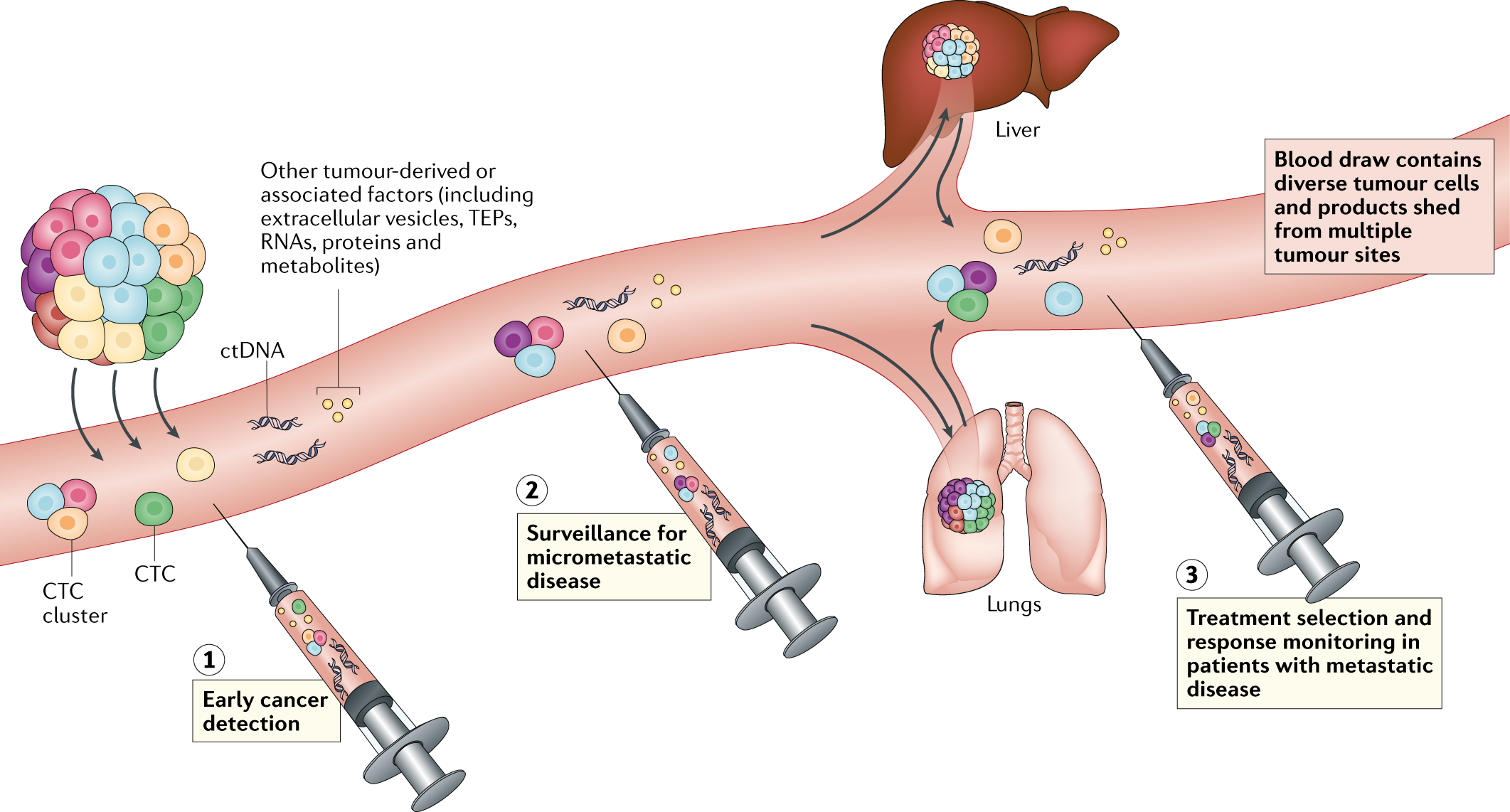
Liquid biopsy enters the clinic — implementation issues and future challenges | Nature Reviews Clinical Oncology

Liquid Biopsy to Identify Actionable Genomic Alterations | American Society of Clinical Oncology Educational Book

Comprehensive genomic profiling in oncology – from vision to reality | Latest news for Doctors, Nurses and Pharmacists | Pharmacy

Liquid biopsy for cancer management: a revolutionary but still limited new tool for precision medicine
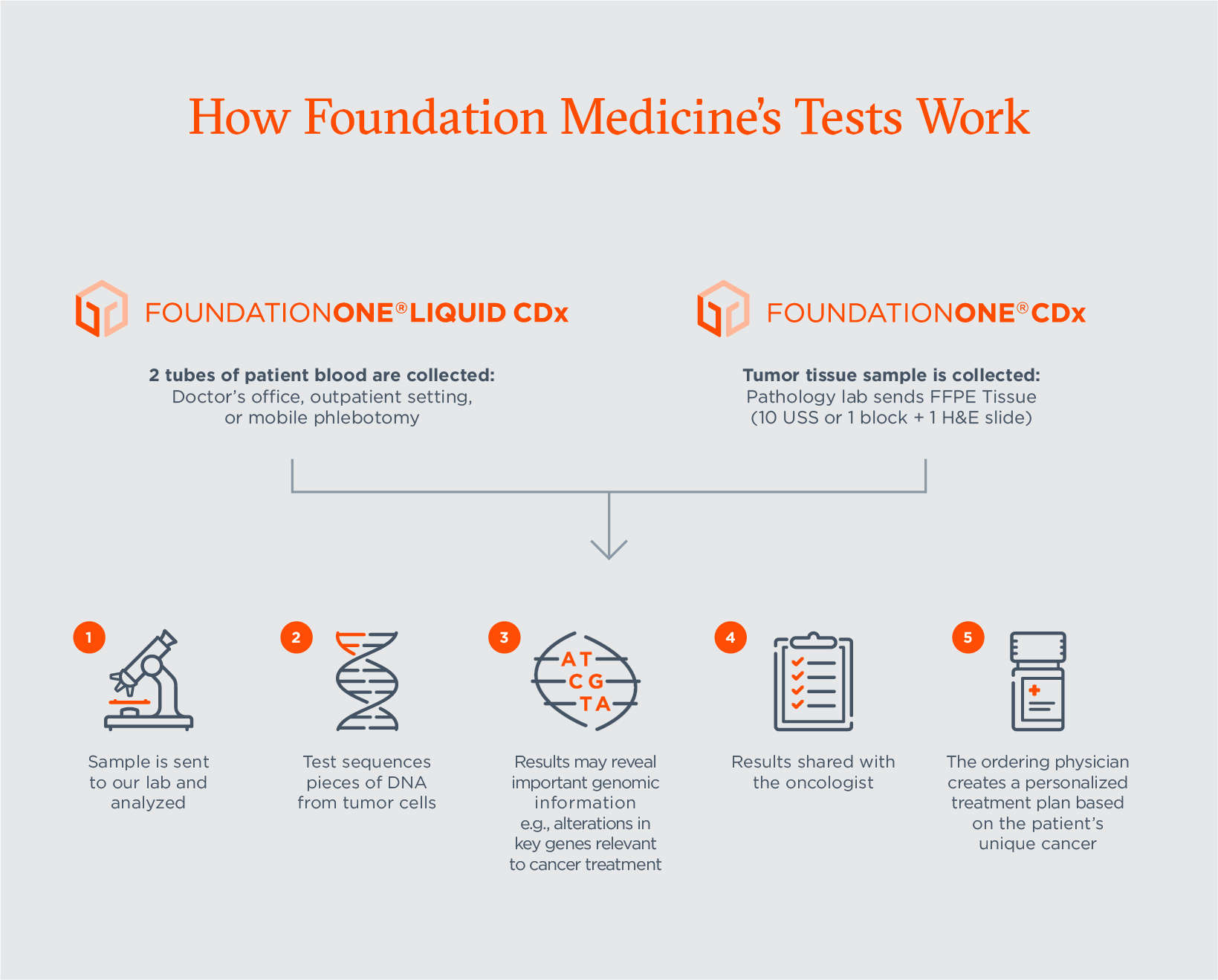
Companion Diagnostics Explained: Their Critical Role in Cancer Care and Our Latest Approvals | Foundation Medicine

Circulating Tumor DNA Associated with Triple-Negative Breast Cancer Recurrence - Pathology - Labmedica.com
Clinical and analytical validation of FoundationOne Liquid CDx, a novel 324-Gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin | PLOS ONE