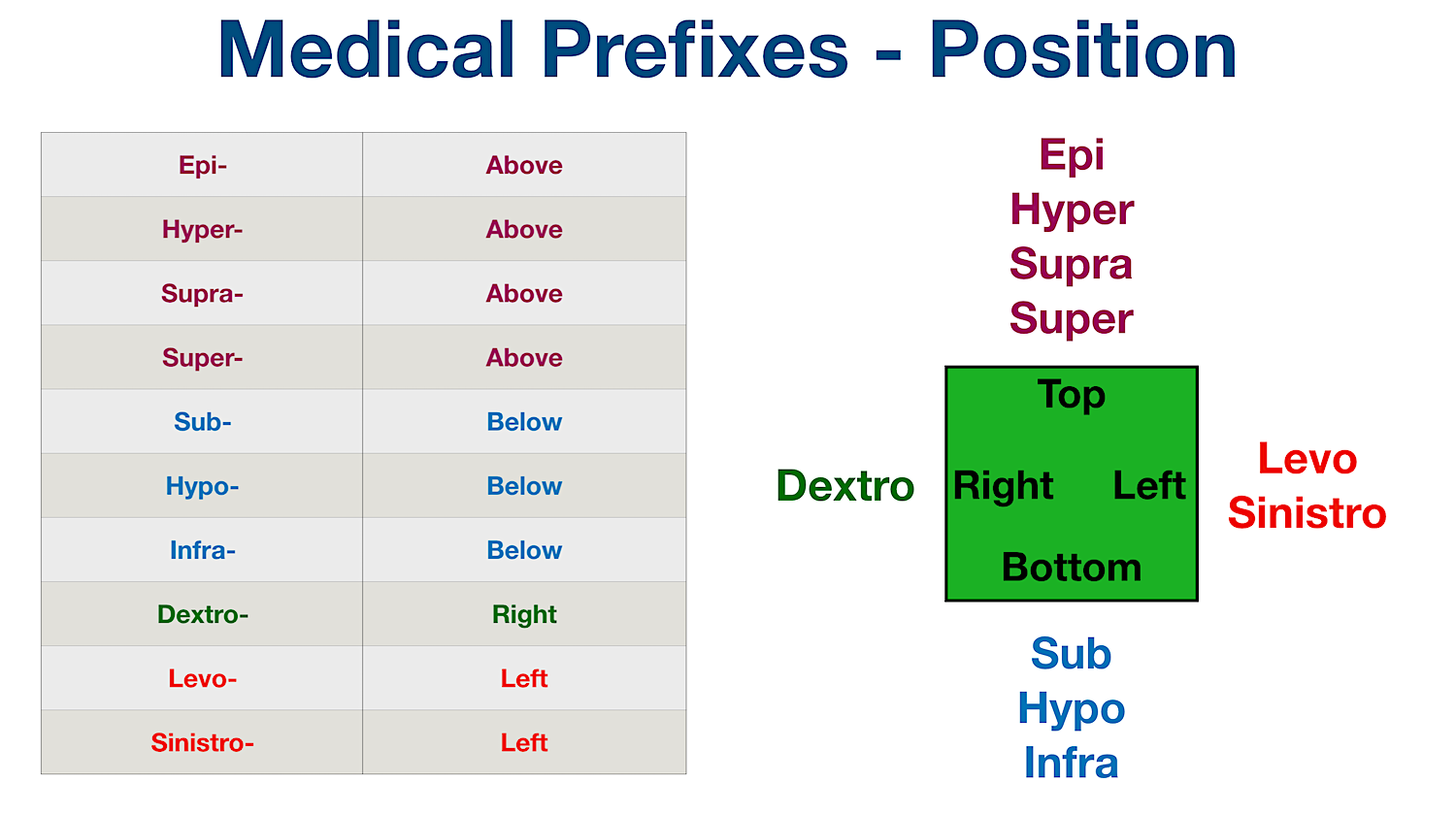
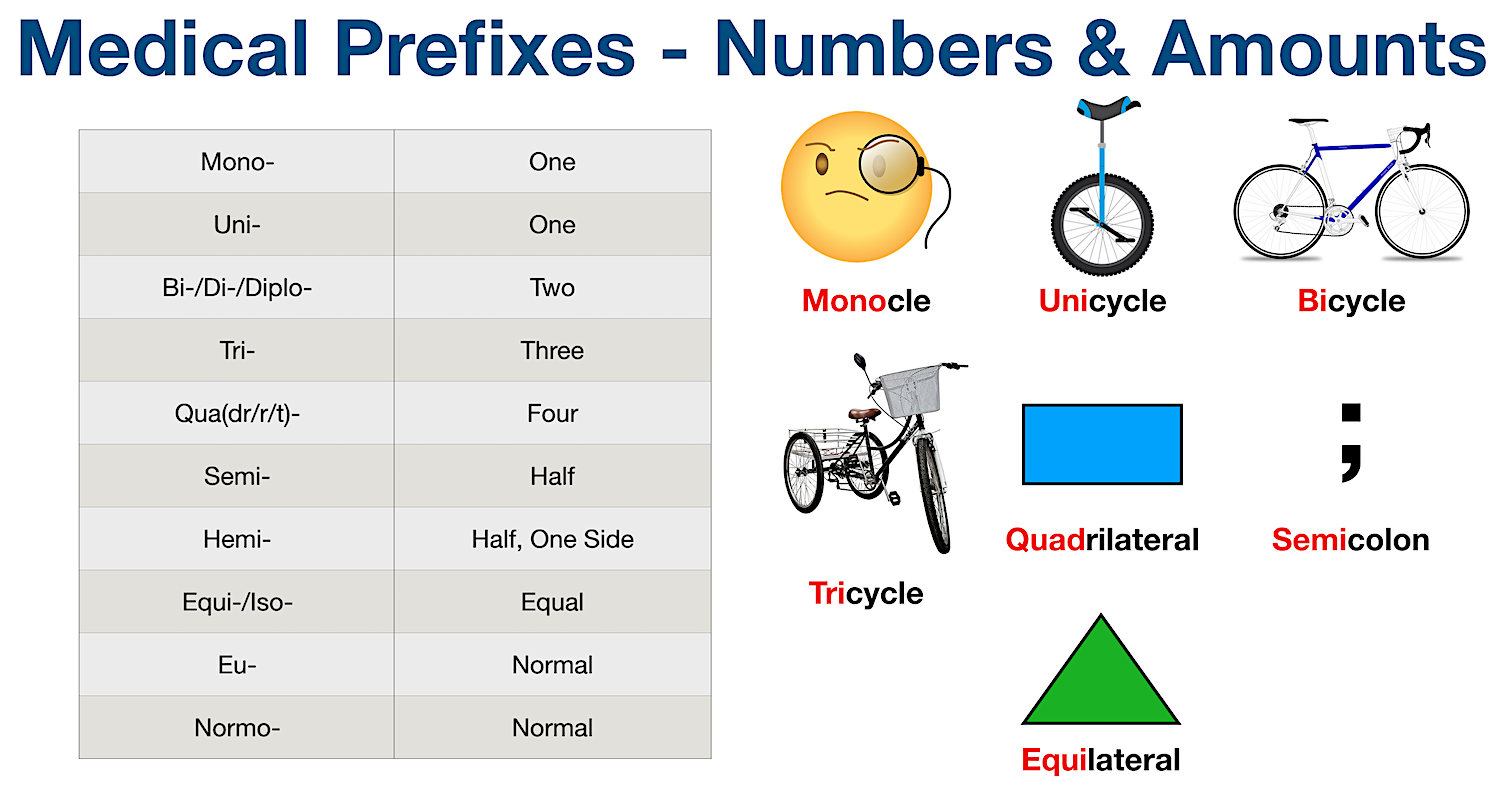
Unit 10 Surgery, Diabetes, Immunology, Lesions, and Prefixes of Numbers and Direction. - ppt download
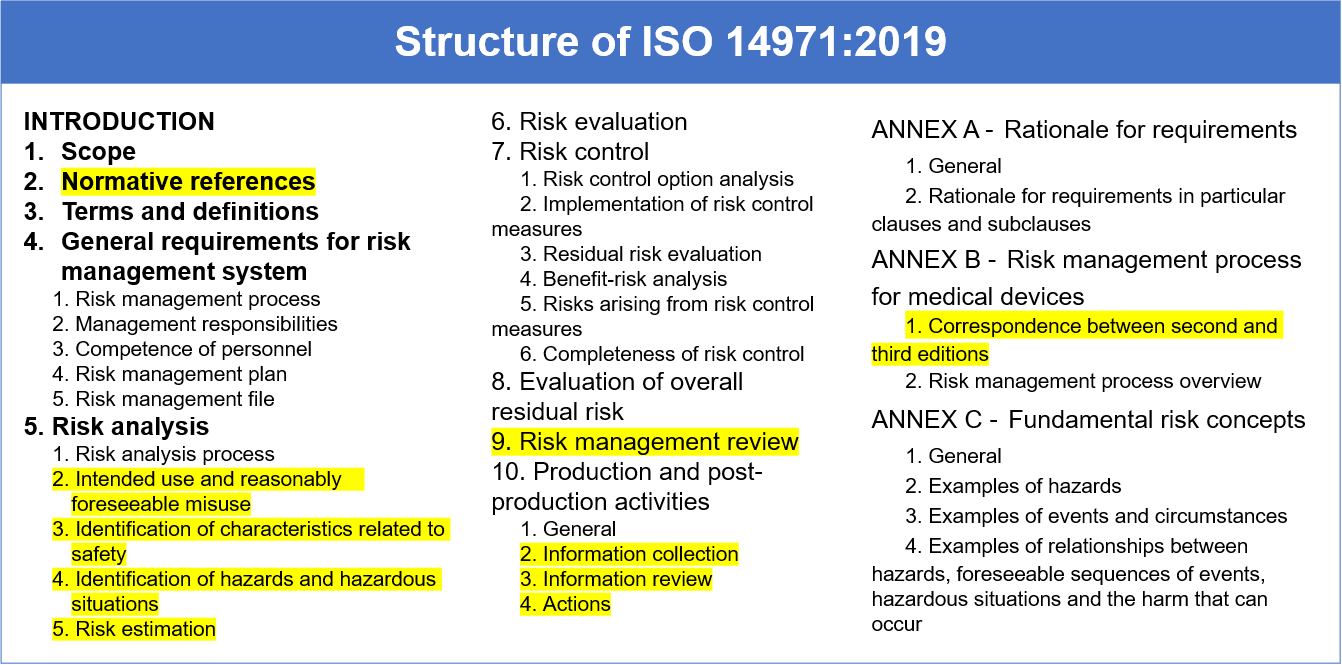
UNE EN ISO 18113-1:2012 In vitro diagnostic medical devices - Information supplied by the manufacturer (labelling) - Part 1: Terms, definitions and general requirements (ISO 18113-1:2009) - European Standards