Polymers | Free Full-Text | Synthetic Approaches for Poly(Phenylene) Block Copolymers via Nickel Coupling Reaction for Fuel Cell Applications | HTML

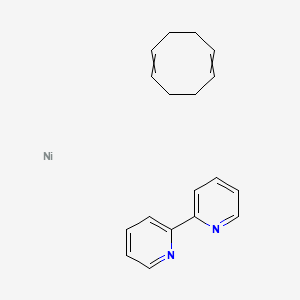
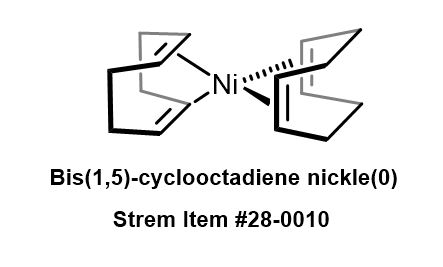
A structural diagram of Ni(COD) 2 . The structure is quasi-tetrahedral... | Download Scientific Diagram
![The Effect of Added Ligands on the Reactions of [Ni(COD)(dppf)] with Alkyl Halides: Halide Abstraction can be Reversible | Organometallic Chemistry | ChemRxiv | Cambridge Open Engage The Effect of Added Ligands on the Reactions of [Ni(COD)(dppf)] with Alkyl Halides: Halide Abstraction can be Reversible | Organometallic Chemistry | ChemRxiv | Cambridge Open Engage](https://chemrxiv.org/engage/api-gateway/chemrxiv/assets/orp/resource/item/60c7584d567dfe0ed4ec6828/largeThumb/the-effect-of-added-ligands-on-the-reactions-of-ni-cod-dppf-with-alkyl-halides-halide-abstraction-can-be-reversible.jpg)
The Effect of Added Ligands on the Reactions of [Ni(COD)(dppf)] with Alkyl Halides: Halide Abstraction can be Reversible | Organometallic Chemistry | ChemRxiv | Cambridge Open Engage

A surprising mechanism lacking the Ni(0) state during the Ni(II)-catalyzed P–C cross-coupling reaction performed in the absence of a reducing agent – An experimental and a theoretical study

Ni(COD)2 coupling of 3,6-dibromocarbazoles as a route to all-carbazole shape persistent macrocycles - ScienceDirect

Ni(COD)(DQ): An Air‐Stable 18‐Electron Nickel(0)–Olefin Precatalyst - Tran - 2020 - Angewandte Chemie International Edition - Wiley Online Library

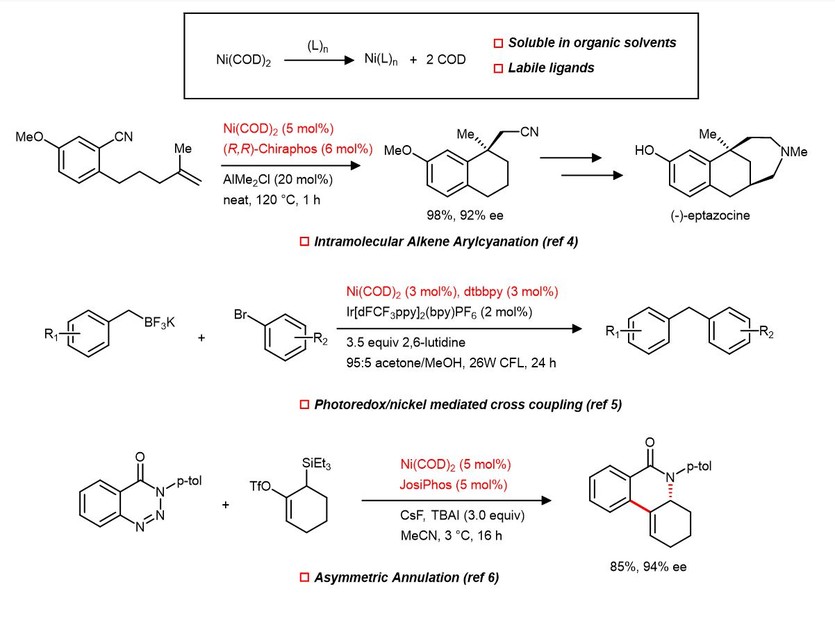
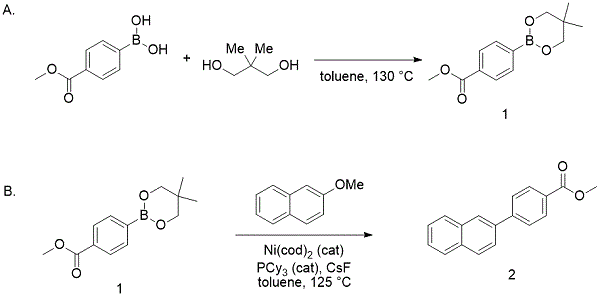
Ni(COD)2/PCy3 Catalyzed Cross-Coupling of Aryl and Heteroaryl Neopentylglycolboronates with Aryl and Heteroaryl Mesylates and Sulfamates in THF at Room Temperature

Synthesis and exchange reactions of Ni-dimine-COD , acetylene and olefin complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C001312A
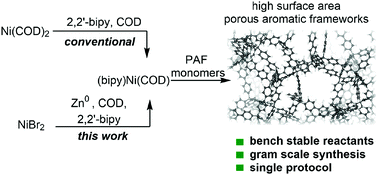
A Ni(COD)2-free approach for the synthesis of high surface area porous aromatic frameworks - Chemical Communications (RSC Publishing)

Synthesis of stabilized Ni nanoparticles from Ni(COD) 2 and dihydrogen | Download Scientific Diagram

Safe and Expeditious Preparation of Ni(cod)2 for Same-Day High-Throughput Screening | Organic Process Research & Development